A mixture consisting of oil in water is an example of the heterogeneous mixture as the oil cannot be mixed in the water and we can easily see them I hope you like my post about "Difference Between Homogeneous And Heterogeneous Mixture" If you want to read more related articles then you can also visit The Best Juices For Memory Homogeneous mixture Heterogeneous mixture Definition Homogeneous mixture is the combination of two or more pure substances so uniformly that each of the substances is indistinguishable from the other substance The pure substances in the heterogeneous mixture are not evenly distributed and result in the formation of a nonuniformA homogeneous equilibrium is one in which all of the reactants and products are present in a single solution (by definition, a homogeneous mixture ) Reactions between solutes in liquid solutions belong to one type of homogeneous equilibria The chemical species involved can be molecules, ions, or a mixture of both For the homogenous reaction

Vihonbtm8o0zpm
Differentiate between homogeneous and heterogeneous mixtures with examples class 9 brainly
Differentiate between homogeneous and heterogeneous mixtures with examples class 9 brainly- The homogeneous mixture is only in the one phase of matter, whereas heterogeneous mixture is always in two or more than two different phases of matter Solutions are termed as the homogeneous mixtures, on the other hand, suspensions or colloids are termed as the heterogeneous mixtures A pure substance is a form of matter that has a fixed chemical composition and a distinct characteristic while a homogeneous mixture is a mixture of two or more compounds with compositions that are uniform or mixed together in such a way that they are indistinguishable from each other Is gold alloy homogeneous or heterogeneous?




Vihonbtm8o0zpm
Despite being a part of the mixture The mixtures are divided into two main categories known as homogeneous mixtures and heterogeneous mixtures The Homo and Straight terms represent the most important difference between homogeneous and heterogeneous mixtures The Homo prefix refers to uniformity while straight indicates nonuniformity A homogeneous mixture is one in which its components are mixed in such a way that it is impossible to differentiate them with the naked eye, being uniformly distributed This type of mixture is in a phase (state of matter) and is also known as a solution or solution An example of a homogeneous mixture or solution is a hot cup of coffeeThis chemistry video tutorial explains the difference between homogeneous and heterogeneous mixtures within the subtopic of the classification of matter It
A heterogeneous mixture consists of two or more phases When oil and water are combined, they do not mix evenly, but instead form two separate layers With that being said, what is the difference between a homogeneous mixture and a heterogeneous mixture quizlet?What is the difference between heterogeneous and homogeneous mixture Define the mixture Define homogeneous mixture Date examples of homogeneous mixtures How do you like coffee? Homogeneous and heterogeneous are two types of mixtures that are studied and apart of science To start identifying the difference between these two mixtures, we can look at the prefixes of each word The prefix homo means single In relation to a homogeneous mixture, this means the mixture only goes through one phase
In this animated lecture, I will teach you the concept of mixture, different types of mixture, homogeneous mixture, heterogeneous mixture, difference between Question Difference between homogeneous and heterogeneous mixture with examples Answer Homogeneous Mixture A mixture in which its different constituents are mixed uniformly (uniform composition); What is the difference between heterogeneous and homogeneous?




Difference Between Homogeneous And Heterogeneous Homogeneous Vs Heterogeneous




Solutions And Mixtures Flashcards Quizlet
Difference between Homogeneous and Heterogeneous Mixture Homogeneous mixture Heterogeneous mixture It can't be separated out physically It can be separated out physically 'homo' means the same 'hetero' means different Example a mixture of alcohol and water Example a mixture of sodium chloride and sand Differentiate between Homogeneous and Heterogeneous mixture with examples by Jaishree Gorane Leave a Comment List the points of difference between Homogeneous and Heterogeneous mixtures5 rows The components of homogeneous mixtures are not physically distinct A heterogeneous mixture




Homogeneous Vs Heterogeneous Mixtures Difference And Comparison Diffen




Homogeneous Mixture Definition Examples Tutors Com
Homogeneous Mixture Definition A homogeneous mixture is a mixture of substances blended so thoroughly that you cannot see individual substances Every sample of the mixture will show the same amounts of each substance Homogeneous mixtures can be solid, liquid, gas, or plasma mixturesDifference between compound and homogeneous mixture learning objectives explain the difference between a pure substance and a mixture explains the difference between an element and a compound explains the difference between a homogeneous mixture and a heterogeneous mixture a useful way to organize our understanding of matter is to think of aStart studying Heterogeneous vs Homogeneous Mixtures Learn vocabulary, terms, and more with flashcards, games, and other study tools
/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



Heterogeneous Vs Homogeneous Mixtures




Interesting Chemistry Difference Between Homogeneous And Heterogeneous Mixture Chemical Chemistrynotes Chemistry Facebook
6 rows In homogeneous mixtures, there seems to be only one component (solute and solvent), but in The key difference between homogeneous and heterogeneous is that homogeneous materials and mixtures have the same uniform composition and properties throughout whereas heterogeneous materials and mixtures do not have either uniform composition or uniform properties Reference 1 Helmenstine, Anne Marie, PhDTherefore, the individual components cannot be separately identified




9th Class Chemistry Differentiate Between Homogeneous And Heterogeneous Mixture Brainly In




Heterogeneous And Homogeneous Mixture Differences Videos Examples
Example 1 Mixture of Oil and Water is a heterogeneous Mixture We are able to see oil and water clearly separately in the mixture Example 2 Mixture of Salt (Sodium Chloride) and Iron filings is a heterogeneous mixture The particles of salt and Iron filings can be seen and distinguished easily Difference between Homogeneous and Heterogeneous mixtures8 Difference Between Homogenous And Heterogeneous Mixture With Examples In physical chemistry and material science, a mixture is the physical combination of two or more substances in which the identities are retained and are mixed in the form of solutions, suspensions and colloids One major characteristic of mixtures is that they can be separated into their components, since each part of the mixture has not reacted with another part of the mixtureExamples of heterogeneous mixtures include sand, oil and water, and chicken noodle soup What is homogeneity and heterogeneity?
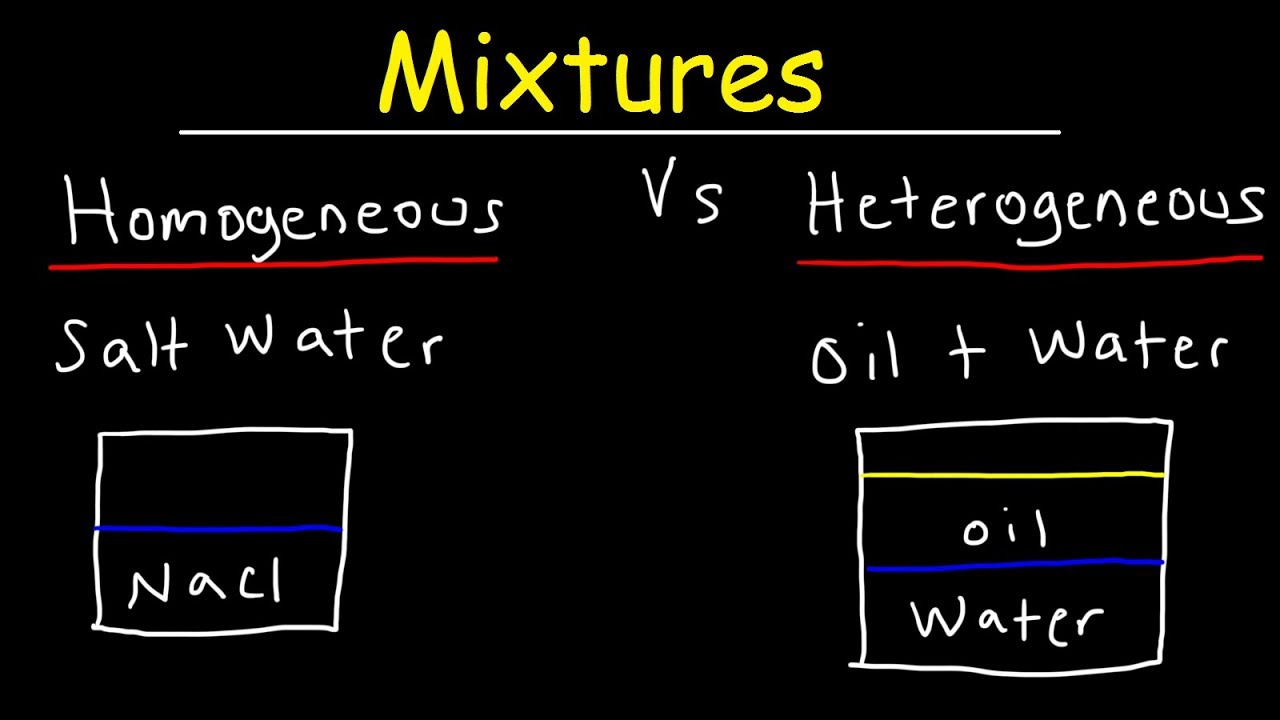



Homogeneous And Heterogeneous Mixtures Examples Classification Of Matter Chemistry Youtube
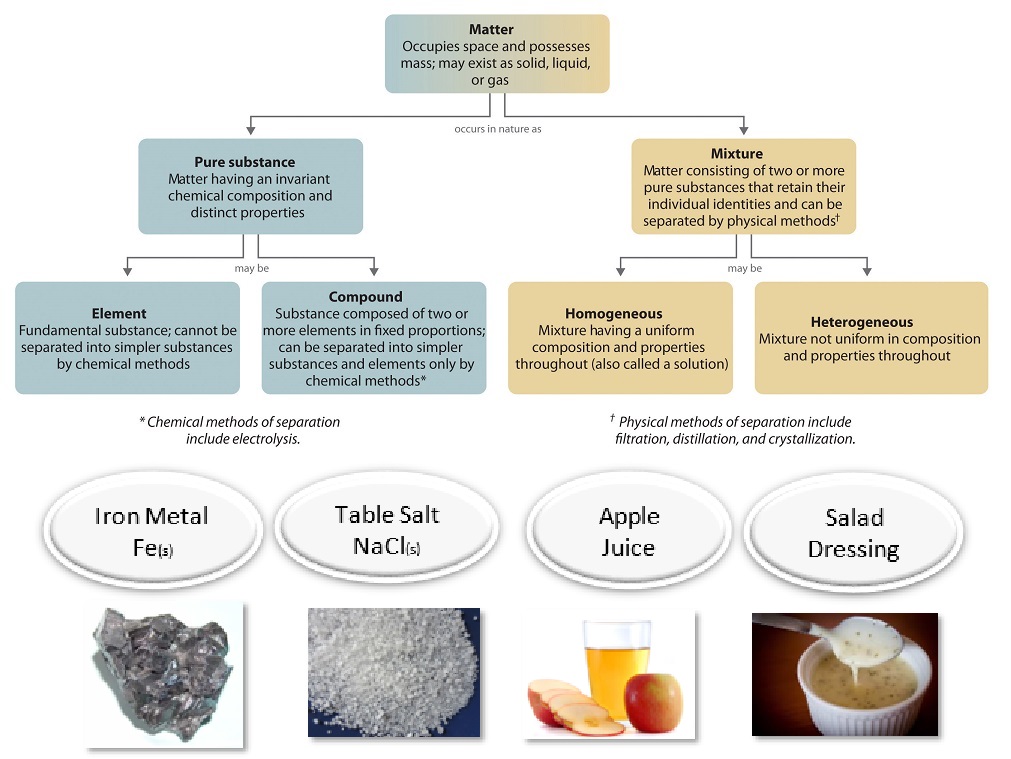



3 4 Classifying Matter According To Its Composition Chemistry Libretexts
Heterogeneous mixtures are not uniform in composition Examples would be wood or bloodCorrect answers 2 question ⚠⚠need ⚠⚠ difference between homogeneous and heterogeneous mixture ⬅⬅ full explanation with examples don't spam Some examples of homogeneous mixtures are most alloys, seawater, brass, vinegar, air, blood, natural gas, etc Heterogeneous Mixture Salt and pepper together make a heterogeneous mixture




Vihonbtm8o0zpm



Mixture
Explain the difference between a homogeneous mixture and a heterogeneous mixture A useful way to organize our understanding of matter is to think of a hierarchy that extends from the general and Smog is another example of a heterogeneous mixture This turbid collection of pollutants can be a mixture of water and contaminants from the If a heterogeneous mixture were divided in two, an unequal amount of all the substances that compose it would remain Examples of heterogeneous mixtures sand, rocks and water, water and oil If water and sand are placed in a container, the latter sinks to the bottom In conclusion, homogeneous and heterogeneous mixtures are totally oppositeExamples of homogeneous mixture A glass of lemonade (mixture of water, lemon juice, sugar, salt) is a homogeneous mixture because the dissolved sugar, salt, and lemon juice are evenly distributed throughout the entire sample You can't easily separate the lemon juice from the water;
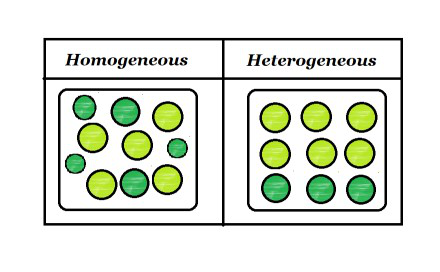



Homogeneous And Heterogeneous Mixtures Geeksforgeeks
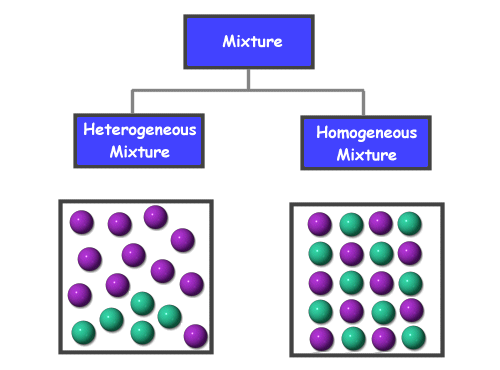



Chemistry For Kids Chemical Mixtures
Homogeneous vs heterogeneous The difference between homogeneous mixtures and heterogeneous mixtures is a matter of scale the heterogeneous mixture can be seen on beaches where sand included many particles like coral, shells and organic matter, etc they all can be separated easily hence known as a heterogeneous mixture but when we take a large amount of sand, it's impossible to separate all the matter, which terns as a homogeneous mixture example Unlike homogeneous mixtures, heterogeneous mixtures do not have the same composition throughout Oil and water is a fine example of a heterogeneous mixture When oil is mixed with water, the separation of two layers is visible to the eye The following picture shows this phenomenon clearly Examples of Heterogeneous Mixtures Here is ur answer Difference between homogeneous and heterogeneous mixture 1) A homogeneous mixture has a uniform composition throughout its mass whereas a heterogeneous mixture has a non uniform composition 2) A homogeneous mixture does not contain physically distinct parts whereas a heterogeneous mixture contains physically distinct




List The Points Of Difference Between Homogeneous And Heterogeneous Mixtures Brainly In




Examples Of Heterogeneous Mixtures Types Made Simple
Homogeneous mixtures have the same composition throughout the system, and heterogeneous mixtures have the opposite The particles in the odd one are arranged randomly while the particles in a homogeneous mixture are arranged uniformly, giving rise to6 rows Through combining two or more substances, a mixture is produced A homogeneous solution tends to Similarly, you may ask, what is difference between homogeneous and heterogeneous?




List The Point Of Difference Between Homogenous And Heterogenous Mixtures




List The Point Of Differences Between Homogeneous And Heterogeneous Mixture Brainly In
Particle size distinguishes homogeneous solutions from other heterogeneous mixtures Solutions have particles which are the size of atoms or molecules too small to be seen A colloid is a homogeneous solution with intermediate particle size between a solution and a suspension Salt and water, for example, are homogeneous mixtures, as is sugar plus water As described by the dictionary of Chemistry, a heterogeneous mixture is a combination in which the constitution is not regular and smooth The elements are not homogeneous in their composition The components can't be dissolved readily Mixtures can be mainly divided into two as homogeneous mixtures and heterogeneous mixtures A heterogeneous mixture has two or more phases and the components can be individually identified A homogeneous mixture is uniform;



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples




Difference Between Homogeneous And Heterogeneous Equilibrium Compare The Difference Between Similar Terms
Basically we subdivide a mixture in homogeneous and heterogeneous mixtures In simple words, in a homogeneous mixture, it is not possible to easily differentiate between its components The components in a heterogeneous mixture can be easily differentiated An example of a homogeneous solution can be a salt solutionSolution Homogeneous Mixtures Heterogeneous mixtures The elements and compounds are uniformly mixed The elements and compounds are not uniformly mixed The properties of the mixture are the same in all compositions The properties differ at different compositions Copper and Zinc are two solids which make brass, which is solidMany people enjoy a cup of coffee at some point during the day Some can drink black, while others can put cream (or some milk substitute) and sugar in their coffee
/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



Heterogeneous Vs Homogeneous Mixtures




Homogenous And Heterogenous Mixtures Chemistry For Non Majors
Whether a mixture is heterogeneous or homogeneous depends on how closely you examine it Sand may appear homogeneous from a distance, yet when you magnify it, it is heterogeneous Examples of homogeneous mixtures include air, saline solution, most alloys, and bitumen Examples of heterogeneous mixtures include sand, oil and water, and chicken• Apple juice is homogeneous • Orange juice with pulp is heterogeneous • Chocolate dough is homogeneous • Italian salad dressing is heterogeneousBy definition, a pure substance or a homogeneous mixture consists of a single phase A heterogeneous mixture consists of two or more phases When oil and water are combined, they do not mix evenly, but instead form two separate layers Each of the layers is called a phase
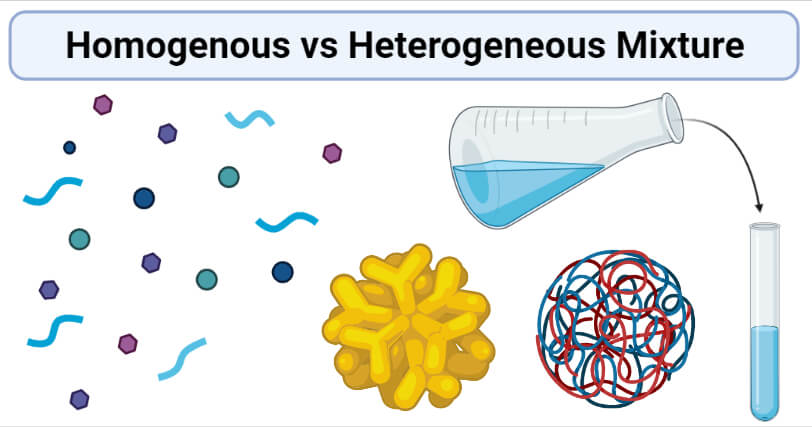



Homogenous Vs Heterogeneous Mixture Definition 8 Key Differences Examples
/definition-of-heterogeneous-mixture-and-examples-605206_final23-ecfa4da6517640429448462eae1f09f7.png)



Definition Of Heterogeneous Mixture With Examples
Difference between Homogeneous and Heterogeneous Mixtures 1 Mixtures that have uniform composition Mixtures that do not have uniform composition throughout 2 Boundary of separation could not be seen Boundary of separation of constituent particles is clearly visible 3 Particles are not indistinguishable If you look closely at sand from a beach, you can see the different components, including shells, coral, sand, and organic matter It's a heterogeneous mixture If, however, you view a large volume of sand from a distance, it's impossible to discern the different types of particles The mixture is homogeneousA homogeneous mixture has the same uniform appearance and composition throughout Many homogeneous mixtures are commonly referred to as solutions A heterogeneous mixture consists of visibly different substances or phases Solutions have




Homogeneous Mixture Definition Examples Tutors Com




What Is The Difference Between Homogeneous And Heterogeneous Mixture
It has no visible boundaries of separation between A homogeneous mixture is a mixture having a uniform composition throughout the mixture For example salt in water, sugar in water, copper sulphate in water A heterogeneous mixture is a mixture having a nonuniform composition throughout the mixture For example sodium chloride and iron fillings, salt and sulphur, oil and waterThe terms homogeneity and heterogeneity are used to describe the uniformity and regularity in spatial distribution of geomaterial properties in
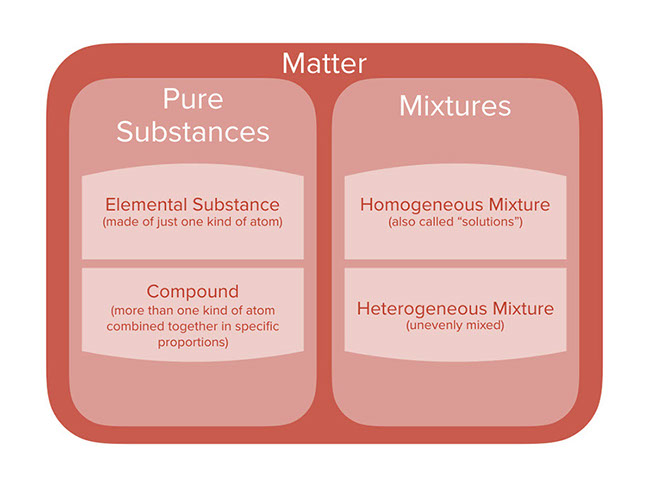



Lesson Categories Of Chemicals And Mixtures




Homogeneous Mixture Example Food
For example, a salad Unlike homogeneous mixtures, in heterogeneous mixtures it is very easy to identify, even with the naked eye, what are the different components that make them up This makes it much easier to separate these mixes at the sameHomogeneous mixture Heterogeneous mixture 1) These are called as solutions These are



Homogeneous Heterogeneous Mixture Definition Examples Selftution




What Is A Homogeneous Mixture Definition And Examples




Heterogeneous Vs Homogeneous Mixtures




Homogeneous And Heterogeneous Mixture Difference Between Homogeneous And Heterogeneous Mixture Youtube




What Is Difference Between Heterogeneous And Homogeneous Brainly In




Give Three Differences Between Homogeneous Mixture And Heterogeneous Mixture Brainly In




Explain The Difference Between A Homogeneous And Heterogeneous Mixture Give An Example For Each Brainly Com




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii



1




Homogeneous Mixture Definition Examples Tutors Com



Sxxv4y Ycx9hem



Http Punainternationalschool Com Assets Upload Ck Images Class ix chemisty july aug material Pdf



Difference Between Homogeneous And Heterogeneous Welding




8 Difference Between Homogenous And Heterogeneous Mixture With Examples Viva Differences
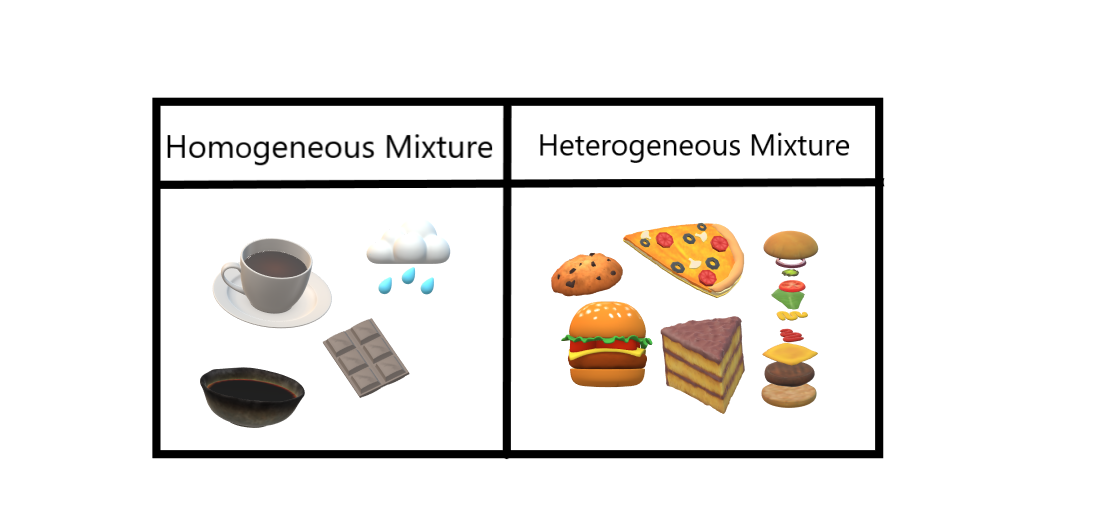



Homogeneous And Heterogeneous Mixtures Geeksforgeeks




Pure Substances And Mixtures Homogeneous Mixture Heterogeneous Mixture Elements Compounds And Mixtures




Homogenous And Heterogenous Mixtures Chemistry For Non Majors




Ppt Objective I Will Distinguish Between Homogeneous And Heterogeneous Mixtures Powerpoint Presentation Id




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Difference Between Homogeneous And Heterogeneous Mixtures Homogeneous Vs Heterogeneous Youtube




Difference Between Homogenous And Heterogenous Mixtures Youtube




Difference Between Homogeneous Mixture And Heterogeneous Mixture Youtube




Ppt Homogeneous And Heterogeneous Mixtures Powerpoint Presentation Free Download Id
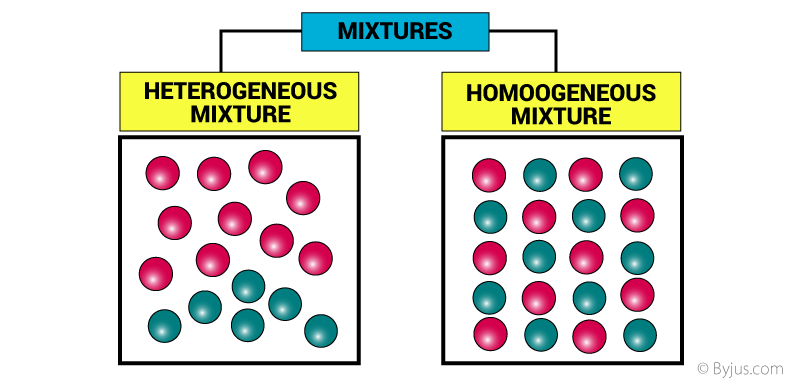



What Is A Mixture Definition Properties Examples Types With Videos




Differentiate Between Homogeneous And Heterogeneous Mixtures With Examples Brainly In




List The Point Of Difference Between Homogeneous And Heterogeneous Mixture Science Is Matter Around Us Pure Meritnation Com




Mixtures Youtube




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii
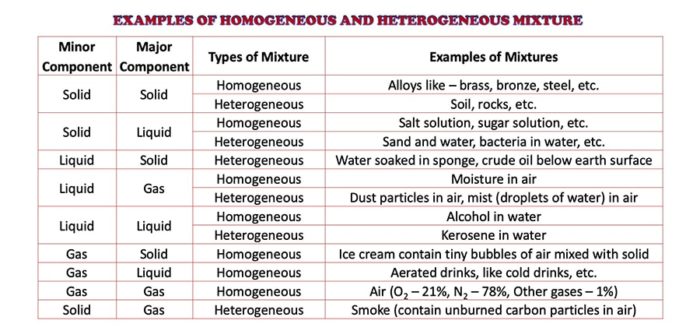



Homogeneous Heterogeneous Mixture Definition Examples Selftution




Q2 Differentiate Between Homog Lido




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Difference Between Homogeneous And Heterogeneous Compare The Difference Between Similar Terms
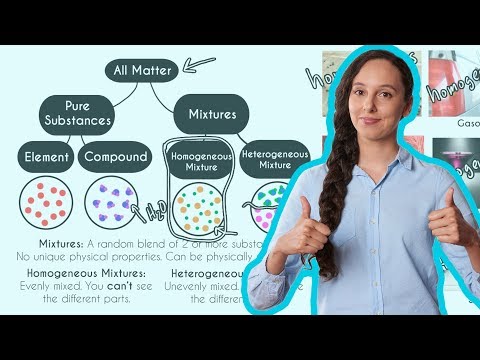



Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Difference Between H Mixture And Heterogeneous Mixture Brainly In




Homogeneous Mixture Examples In Daily Life



1



Http Punainternationalschool Com Assets Upload Ck Images Class ix chemisty july aug material Pdf



3




Heterogeneous And Homogeneous Mixture Differences Videos Examples




Homogeneous Mixture Example




What Are Some Examples Of Homogeneous Mixtures And Heterogeneous Mixtures Enotes Com
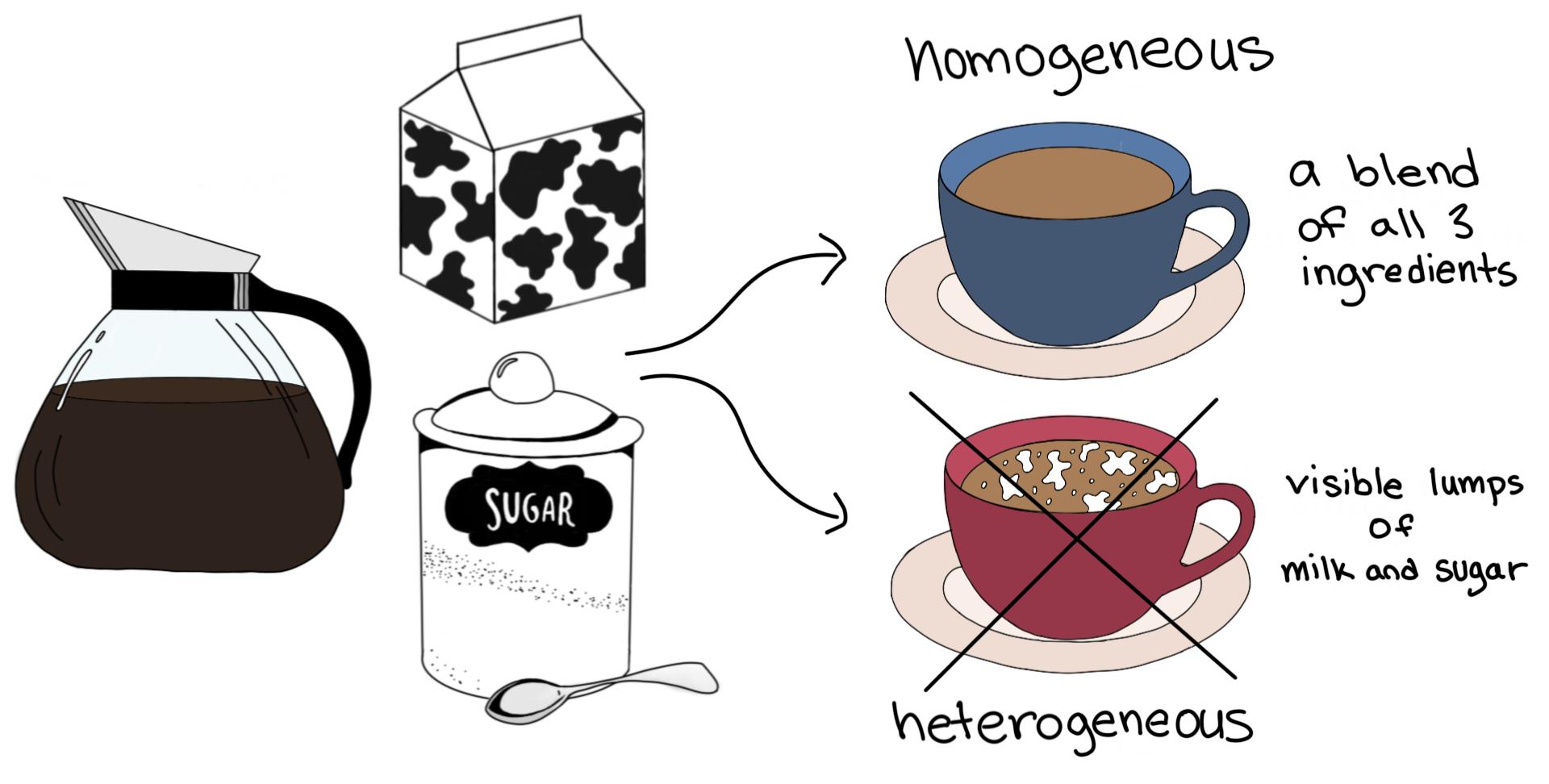



Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Mixtures And Solutions Cpd Rsc Education




Difference Between Homogeneous Mixture And Heterogeneous Mixture




Homogeneous And Heterogeneous Mixture Difference Between Homogeneous And Heterogeneous Mixture Youtube
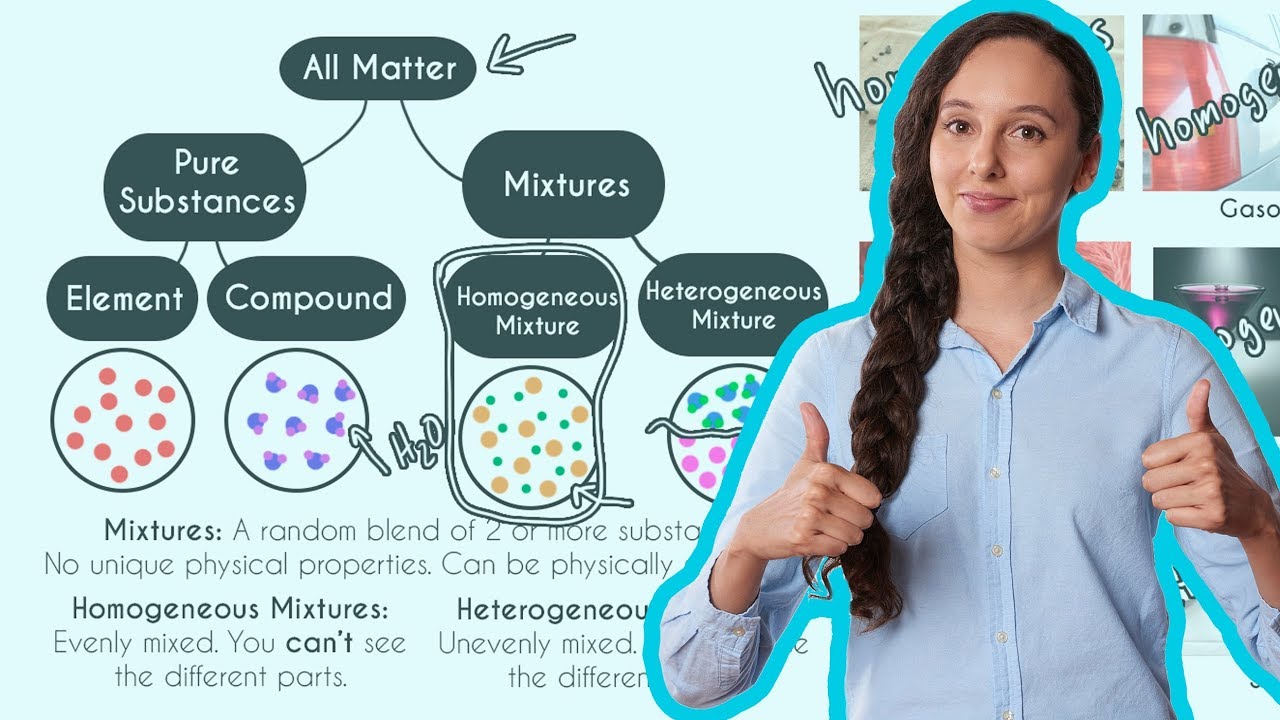



Homogeneous And Heterogeneous Mixtures Youtube




What Is The Difference Between Heterogeneous Mixture Vs Homogenous Mixture Brainly Com




Differentiate Between Homogeneous And Heterogeneous Mixtures With Examples Youtube



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Homogenous Definition And Examples Biology Online Dictionary




1 Differentiate Between Homogon Eous Aid Heterogeneous Mixt Scholr




What Is A Heterogeneous Mixture Definition And Examples




Differentiate B W Homogeneous And Heterogeneous Mixtures Teachoo




Homogenous Compounds And Mixtures Homogeneous Mixture Heterogeneous Mixture



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples




Difference Between Homogeneous And Heterogeneous Compare The Difference Between Similar Terms




Give 8 Differences Between Homogeneous Mixture And Heterogeneous Mixture Brainly In




Heterogeneous Mixture Lesson For Kids Definition Examples Video Lesson Transcript Study Com
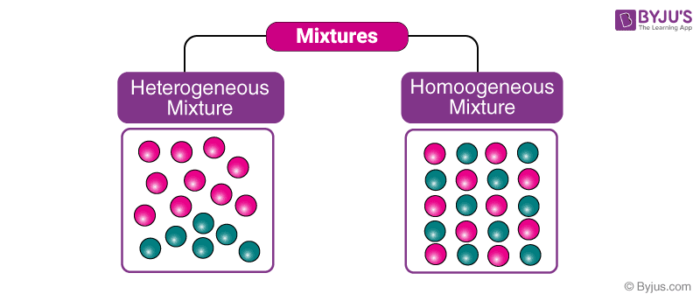



Heterogeneous And Homogeneous Mixture Differences Videos Examples




What Is The Difference Between Homogeneous And Heterogeneous Mixtures



Mixture




Homogeneous Mixture Examples In Kitchen




Classification Of Matter Chemistrygod



3




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




List The Points Of Differences Between Homogeneous And Heterogeneous Mixtures
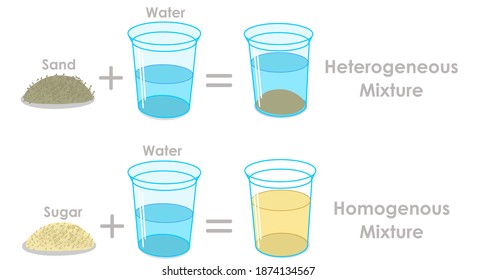



Mixtures Images Stock Photos Vectors Shutterstock




Homogeneous And Heterogeneous Mixtures Heterogeneous Mixture Venn Diagram Examples Venn Diagram Template




Homogeneous And Hetrogeneous Mixtures Definition Examples Teachoo




10 Examples Of Mixtures




Compound Vs Mixture Difference And Comparison Diffen




What Do You Need To Know About Heterogeneous And Homogeneous Mixtures




Homogeneous And Heterogeneous Mixture Difference Between Homogeneous And Heterogeneous Mixture Youtube



Mixture



0 件のコメント:
コメントを投稿